Taking Account of the U.S. Navy Scrubber Test Cheating
Using Source Test Data to Determine Correction Factors to Compensate for Cheating
During the following chrome plating scrubber emissions source tests listed in Section B below Mare Island engaged in a lot of cheating to obtain artificially low chromic acid emission test results in a desperate attempt to retain the plating shop permits to operate an air pollution abatement prevention device. In order to determine and calculate an accurate actual normal conditions chromic acid emission rate for the scrubbers it is necessary to consider the methods of cheating used. Then, data from the different tests of the same scrubbers can be compared and contrasted mathematically to obtain good and accurate correction factors to be used to adjust the test results to obtain results that would have been measured if there had been no cheating.
This is a page in progress. It will be referenced on the page, currently in preparation, that reports best estimates of the amount of chromic acid discharged by the Mare Island Plating Shop. The calculation factors on this page will be used in the calculations provided on that page. In the meanwhile if you would like to see the dangerous amount of chromic acid emitted by the scrubbers, as is estimated by corrected perjerous calculations made by U.S. Navy Managment you can use this link: PERJURY.
If you would like to review the Scrubber Discharge Calculations Page in progress you can use this link: Scrubber Discharges
A. The Following Links will lead you directly to the applicable section for each of the calculated correction factors:
1. Scrubber Efficiency Correction Factor: SECF
2. Hard Chrome Plating Bath Correction Factor: HBCF
3. Decorative Chrome Correction Factor: DBCF
4. Anodizing Bath Correction Factor: ABCF
B. The Available Tests and Data:
There are three sets of tests of the two Mare Island Shipyard chrome plating scrubbers. These tests were as follows:
1. 1987: BAAQMD Supervised Tests. From this test set the only data available is total scrubber efficiency for each of the two chromium scrubbers.
2. April 5, 1988 BAAQMD Supervised Test: This was a test of the two hard chrome plating tanks and their associated scrubber. A complete data set is available from this test.
3. April 6, 1988 BAAQMD Supervised Test: This was a test of the Decorative chrome plating tank, the aluminum anodizing tank and their associated scrubber. A complete data set is available for this test.
4. August 10, 1988 RSA Conducted Test of the hard chrome plating tank and its associated scrubber. A complete data set is available from this test.
5. August 17, 1988 RSA Conducted Test of the decorative chrome plating tank, the aluminum anodizing tank and their associated scrubber. A complete data set is available for this test.
Three Sets of data are also available that are necessary for calculation of the necessary Correction Factors:
1. Mare Island Naval Shipyard Toxic Inventory Preview of 1989
2. Transcript of Telephone Conversation with the Harrington Scrubber Technical Representative 1996
3. The Mare Island Naval Shipyard Plating Shop Process Instruction Part A and Part B.
The data contained in all of these test documents and other sources are used to determine the necessary correction factors to be applied to the data to calculate the actual amount of chromic acid that was routinely discharged into the air from the plating shop during the years of 1951 to 1989 when the plating shop was finally permanently closed.
C. The U.S. Navy Committed Perjury in Writing During The Plating Shop Arbitration Hearing In August 1988
The U.S. Navy was so determined to deny the people it had murdered, injured and crippled their legal rights to medical care and compensation under federal law as a result of their being deliberately exposed to toxic amounts of chromic acid discharged out of the plating shop for decades that it was willing to commit deliberate Perjury in writing.
The Mare Island Union President at the time of the time of the closing of Mare Island in 1996 had been severely poisoned by the plating shop chromic acid spill on February 17, 1988. His health and strength had been permanently severely diminished by this, but the spill had been kept a closely guarded secret from everyone and so he had no idea of what had happened to him. Shortly before the Shipyard closed he accidentally found a copy of the Official Critique Report of this spill and realized immediately that it was the cause of his health problems. Then, he went on a trip around the shipyard and talked to as many of the people his people his Union represented. During these conversations a majority of these people remarked to him that they had once been in great health, but after coming to Vallejo they did not feel well most of the time. Then, he went on a hunt for papers and found many you see posted on this web site. Finally he had a confrontation with the Deputy Assistant Head of the Occupational Safety and Health Department who flatly denied there had ever been a problem with the plating shop chromic acid scrubbers. This lie mandated that something be done and so a Union Grievance was filed against the Shipyard asking for the back pay of the hazardous duty pay for chemical exposure due to those who had been exposed to chromic acid. There were a great many difficulties in this effort and he was subjected to many hardships and schemes, and the effort finally culminated in a hearing before an Arbitrator in August 1996.
During this hearing evidence was submitted to the Arbitrator. When evidence is submitted in a Grievance Arbitration Hearing it is under oath that it is the truth, the whole truth, and nothing but the truth. Management was represented by the Shipyard Attorney and so had no reason to misunderstand this. During the hearing Management submitted Management Exhibit No. 11. This was an "analysis" purporting to show a "worst case" and "best case" result from professional engineering calculations that demonstrated conclusively the hard chrome and decorative chrome/aluminum anodizing scrubbers could not possibly have emitted harmful amounts of chromic acid. There is no doubt the Arbitrator relied very much on this in making his decision against the Union. Management Exhibit No. 11 is full of big lies, outright lies, partial lies, substitutions and evasions. It is, on its face, the purest possible premeditated and dastardly act of deliberate and willful perjury committed by Management Representatives and Professional Counsel while under oath during the hearing.
This Management Exhibit No. 11 document is the prime and most outstanding true demonstration of the true moral character of the U.S. Navy. You are welcome to view it and an analysis of its twisted character using this link:
U.S. Navy Perjury In Writing
It is of most importance to understand and realize that this document, Management Exhibit No. 11. is not even a proper estimate of the true extent of the poisoning. The document that demonstrates this is still in preparation.
D. The Main Underlying Cheating Technique
The way Mare Island cheated most effectively in all of the tests of chrome plating was to decrease the amount of electrical current (Amperes) passing through the plating bath during the tests. The proof of this is that the Mare Island Plating Shop Instruction, Table 1 and Appendices 1 and 7 specify that when chrome plating is performed the correct amount of electrical current that MUST be applied to the piece of metal that is being chrome plated is 2.5 Amperes per square inch of material being plated. In order to eliminate any argument arising over this chrome plating technical specification it seemed best to demonstrate that during normal operating conditions the personnel at the Mare Island plating shop regularly used 2.5 Amperes per square inch of material being plated.
The two hard chrome plating tanks that were used for the April 5, 1988 and August 10, 1988 RSA scrubber source tests are identified in the test documents to be tanks JFC and JQP. The Dr. Gribbs notes of Appendix III of the RSA Test of August 10, 1988 identifies Tank JFC to be Tank T-5, and Tank JQP to be Tank T-4 as these tanks are identified on the following plating shop chrome plating diagram prepared by the Shipyard:
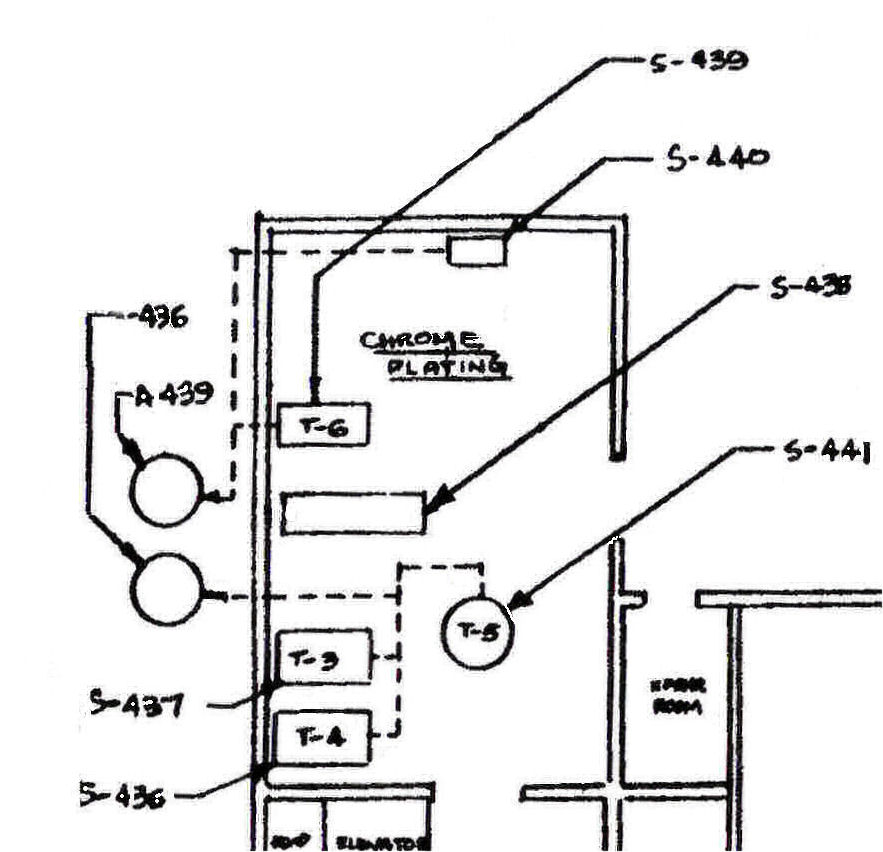
The Mare Island Naval Shipyard Toxic Inventory Preview provides the following Ampere-hours used per chrome plating tank during the partial year of operations in 1988. These are:
1. Tank T-4, Hard Chrome Plating Tank: 1,250,000 Ampere-hours per year.
2. Tank T-5. Hard Chrome Plating Tank: 2,600,000 Ampere-hours per year.
In the Shipyard Commander Letter of March 17, 1992 it is stated the plating shop ceased operation during August of 1988. This means these Ampere-hour per year figures provided in the Toxic Inventory Preview in 1989 indicate only about seven and a half months of use. Also, the plating shop was shutdown for about a month following the airborne spill from the chromic acid scrubbers on February 17, 1988. Adjusting the figures to account for a full year's plating shop use provides:
1. Tank T-4 Full Year = 1,250,000 x 12 months / 6.5 months used = 2,307,692 Ampere-hours per year.
2. Tank T-5 Full Year = 2,600,000 x 12 months year / 6.5 months used = 4,800,000 Ampere-hours per year.
The federal work year is 240 days per year. This allows the calculation of a per-tank daily Ampere-hour electrical usage rate:
1. Tank T-4 Per Day = 2,307692 Ampere-hours per year / 240 days per year = 9612 Ampere-hours per day.
2. Tank T-5 Per Day = 4,800,000 Ampere-hours per year / 240 days per year = 20,000 Ampere-hours per day.
Assuming two shifts per day constituting 12 hours per day with turnover time we can calculate the hourly electrical usage rate:
1. Tank T-4 Hourly = 9612 Ampere-hours per day / 12 hours per day = 801 Amperes per hour.
2. Tank T-5 Hourly = 8666 Ampere-hours per day / 12 hours per day = 1667 Amperes per hour.
Assuming a 2.5 Ampere per square inch electrical current specification actually being used, the size of the piece in the bath being plated can be calculated:
1. Tank T-4 Piece Size = 801 Amperes / 2.5 Amperes per square inch = 320 square inches = 2.22 Square feet.
2. Tank T-5 Piece Size = 1667 Amperes / 2.5 Amperes per square inch = 666 square inches = 4.63 Square feet.
3. Adding the area of both pieces together provides a total square footage being plated of 2.22 + 4.63 = 6.85 square feet.
The capacity of the plating shop to process and prepare material for the chrome plating process needs to be considered. In order for the shop to process 6.85 square feet of material to be continuously plated through both baths simultaneously for an entire year there must be a kind of assembly line preparing pieces at the same time to be continually plated. The Mare Island Plating Shop Process Instruction, Appendices 1 and 7 clearly demonstrate that the many complex processes required to prepare metal for the plating process are so involved, and time consuming that is it virtually impossible for the plating shop to do this. The only reasonable assumption is that the plating shop actually plated far less than 6.85 square feet of metal for 12 hours a day and 240 days per year to justify the actual Ampere-hours per day shown to have actually been used in the Mare Island Toxic Inventory Preview. This provides for only one conclusion and this is that the Mare Island Plating shop not only DID use the 2.5 Amperes per square inch plating current specification but very likely used much more regularly.
E. Cheating in the 1987 BAAQMD Tests
The Big Question answered by this section is: Did the U.S. Navy know that the Mare Island plating shop was emitting harmful and injurious quantities of chromic acid out into the ambient air where innocent victims were exposed to it prior to 1987? The answer is answered by the fact they cheated on the 1987 BAAQMD supervised test. If they had not known that the emissions of chromic acid were out of control and causing deaths, injury and harm, then why all the cheating?
The test document from this test is not available. But data from it has been incompletely cited in the RSA August 30, 1988 test report. The citation of data from other tests in this RSA report allows some understanding of the cheating that took place on this test. This is the RSA report data table that cites the applicable data:
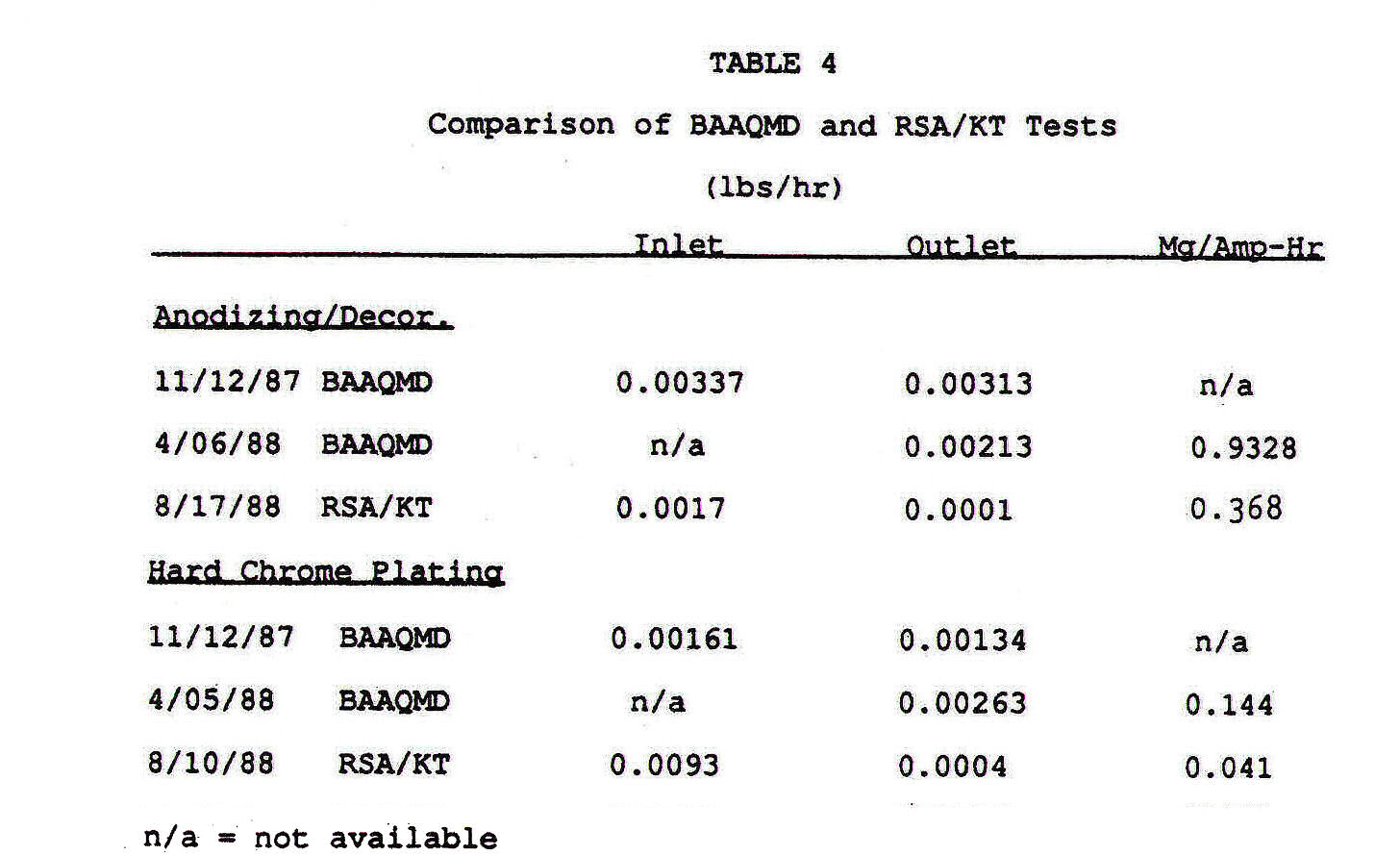
Notice that the annotation "n/a" which usually means "not applicable" is indicated to mean "not available". This means that RSA did not have full and complete access to the BAAQMD 1987 or 1988 supervised tests. This means the U.S. Navy withheld this data from RSA. Also, notice how selective the unavailable data is. I believe this selectivity is designed to ensure there can be no detection of previous cheating and to hide the normal operating bath electrical currents from RSA personnel. I believe that the following can be reasonably be concluded:
1. The missing Mg/ampere-hour data from the 1987 BAAQMD test indicates the hard chrome and decorative chrome/anodizing tests were run at extremely low electrical current values that were far below normal for this test.
2. The missing lbs/hr data for the April 5 and April 6 BAAQMD test is intended to hide the fact that the plating baths were operated at much higher electrical currents than during the RSA tests. This data would have directly contradicted the following statement provided in the RSA report on page 9:

Then there's another illuminating statement provided on Page 18 of the RSA Report:

This statement is meant to help to explain the extremely poor normal operating condition scrubber efficiencies (7% and 17%) discovered during the BAAQMD supervised 1987 tests. But, as is demonstrated shortly, the two hard chrome plating tanks only experienced an electrical current of 415 Amperes during the 1987 BAAQMD test, then they were exposed to a maximum of 8650 Amperes during the April 5, 1988 BAAQMD test, and finally 4,400 Amperes during the August 10, 1988 RSA Test. In the two 1988 tests the fully overhauled and optimized scrubber efficiency was found to be over 93%. The quick and simple answer to this is this statement just doesn't make any sense except from the perception that it is simply a big lie put there to try to detract the mind of the reader from the horror fact that the plating shop scrubbers were, at best only 7% and 17% efficient for effectively all of their operating lifetimes from 1951 to 1988.
Operating two hard chrome plating tanks at 4150 and 4500 Amperes is not an abnormal occurrence. The real problem all this cheating was designed to cover up was that the two chrome plating scrubbers were not maintained at all, they were let to become rocked up, they were operated without water and they were a clear and present danger and major health hazard to the entire shipyard for a very long time causing uncounted deaths and other injuries. The U.S. Navy knew this in 1988 and it fed lies and other falsehoods to RSA so that it would pump out a report that, hopefully, would allow the U.S. Navy to continue poisoning innocent people indefinitely into the future.
Here's what I think happened. The BAAQMD sprung a suprise test of the chrome plating operations at Mare Island as part of its general investigations of plating shops being conducted as part of the process of formulating new chromic acid discharge regulations. Being pressed for time Mare Island could only adjust the plating bath amperage to an artificially low level. The BAAQMD, knowing what they were doing, figured this out and rewarded Mare Island for this cheating with the BAAQMD letter of 1-20-88 that put all of the plating shop permits into jeopardy. But Mare Island knew it was the chrome plating part of the shop the letter was really aimed at and this is why they only rebuilt the two chrome plating scrubbers and the subsequent BAAQMD supervised test on April 5, 1988 test was concerned only with the chrome scrubbers even though the BAAQMD letter demanded a retest of all the scrubbers. Then, during the April 5, 1988 tests Mare Island operated the hard chrome plating tanks honestly with average amperage values of 3880 and 4391 Amperes. It is easy to use the above data to calculate a good estimate of the amount of current used during the 1987 BAAQMD Test:
Current Used during 1981 BAAQMD Test = RSA Amperage x (chrome into scrubber BAAQMD 1987) / chrome into scrubber 1988 RSA)
2400 Amperes X (.00161 lb/hr / .0093 lb/hr) = 415 Amperes
This explains why the BAAQMD came back at Mare Island so strongly and so quickly. It is a ridiculously low value that would have had the appearance of contempt to the BAAQMD regulators.
To sum it up: Here's the sequence of events based on hard chrome plating test Amperes:
1. 1987 BAAQMD Test: 415 Amperes
2. January 1988: BAAQMD test cheating jeopardizes all plating shop permits because of the ridiculous 415 Amperes.
3. April 5, 1988 Test: 8800 Amperes
4. Results of April 5 Test result in plating shop chrome plating permits denied.
5. August 10, 1988 RSA Test: 4400 Amperes
6. Plating shop permits denied initially, but Mare Island agrees to live within the recommendations of the RSA Test, which is impossible to do, and Mare Island gets its permits.
7. Mare Island must perform a Shipyard Wide health risk assessment that it knows will disclose chromic acid based adverse health effects.
8. Mare Island cancels permits rather than perform the health risk analysis.
It is important to note that at the time these tests were conducted in 1987 and 1988 very little was known about the particle size distribution of the chromic acid particles emitted from a working chrome plating bath and how this affected scrubber performance. The Harrington Scrubber Technical Representative stated that the prevailing belief at that time was that hard chrome plating emitted more chromic acid out of the scrubber than did decorative chrome plating. More accurate testing conducted in the 1990's demonstrated that the opposite was the case and that it is decorative chrome plating that is "dirtier" than hard chrome plating. This is the reason the Amperage cheating was greater in the hard chrome baths than in the decorative chrome bath in these tests. It was later discovered that the reason decorative chrome plating is the dirtier process is the decorative chrome plating bath produces droplets so small they tend to escape through a scrubber without being captured.
The extremely high level of cheating in the anodizing bath is due to the fact the anodizing bath emits a very great cloud of mist when it is operating and visibly, it's a horror. There is because of the very high amperage. But the amount of chromic acid actually emitted is much smaller than the appearance and this is because there is about 1/3 the amount of chromic acid dissolved in it (75 grams) to the amount dissolved in a chrome plating bath (250 grams). Also, the droplets generated during anodizing are much larger and are more easily captured by a scrubber. So there was very little reason to cheat so much on this test. But at that time people in the plating world didn't know these things or take them into account.
Finally, there is the smell test. With the April 5, April 6, August 10, and August 17 tests being filled with every possible kind of cheating and manipulation it's just impossible to believe that the 1987 tests are somehow the exception and are pure. If they were so pure then why did the U.S. Navy hide the data from RSA and consequently from everyone else?
F. Cheating By Elimination of a Chromic Acid Source:
The April 5, 1988 and August 10, 1988 scrubber source tests state that they measured the chromic acid emitted from two hard chrome plating tanks. The test documents identify these to be tanks "JFC" and "JQP". In Appendix III of the August 10, 1988 test Dr. Gribbs recorded that Tank "JQP" is actually Tank T-4 as shown on the above diagram, and Tank "JFC" is actually Tank T-5. Tanks T-4 and T-5 were tested on April 5, and August 10, 1988 and Tank T-3 was not included. There is nothing in the record to show why Tank T-3 was not included. It should have been included because The Mare Island Toxic Inventory Preview of 1989 shows that tank T-3 actually used, in the partial year of 1988, 1,300,000 Ampere-hours of electricity, so it was regularly used during normal operations This makes it necessary to account for the output of chromic acid from Tank T-3 that took place during normal operating conditions, in the years between 1951 and 1989 when the plating shop was permanently closed..
The plating shop chrome plating diagram shown above clearly shows tanks T-3, T-4 and T-5 connected to the same scrubber, A-436.
The only thing to conclude is that Mare Island made the commitment to the BAAQMD that Tank T-3 had been permanently put out of commission prior to the April 5, 1988 source tests. However, the data shows that prior to this date the Tank T-3 was extensively used.
The proof that this tank was disconnected from the scrubber is provided by the annotated Mare Island Work Permit that authorized the rebuilding of the two chrome scrubbers prior to the April 5, 1988 Test:

This work permit refers to a "round...tank in the corner". The chrome room diagram above shows a square tank in the corner and a round tank in the center of the room.. This is probably just another little bit of cheating designed to make it difficult to actually identify the identity of the two tanks that were actually tested on April 5, and August 10,1988. Dr. Gribbs hand written notes have a very high degree of confidence and so it has to be understood that Tanks T-4 and T-5 were the tanks actually tested regardless of any minor discrepancy with regard to tank shape shown on the chrome plating room diagram.
The three plating tanks were independently rated for a maximum electrical current of 5000 Amperes each. There is no data that shows the three tanks could not have been operated simultaneously. It must be assumed that during normal operations there were times when all three hard chrome plating tanks were operated simultaneously. The missing chromic acid exhausted from Tank T-3 has to be added to the amounts measured to have been emitted from tanks T-4 and T-5 when they were tested in order to have a more accurate idea of the amount of chromic acid discharged into the breathing air of Mare Island during normal operations of the plating shop.
The amount of chromic acid that will be added to the final scrubber output for missing tank T-3 will be calculated as follows:
Tank T-3 Output = (Corrected Tanks T-4 and T-5 Output) / 2
G. The Reasons Why Correction Factors Are Necessary:
Correction Factors are necessary, one for each scrubber and another for each chrome source, hard chrome and decorative / anodizing, to compensate for two things:
1. The Scrubber Efficiency Correction Factors (SECF): The conditions the two chromic acid scrubbers were operated under were the worst possible. They were not maintained or monitored, they were not provided with water flow and so they were very inefficient at capturing chromic acid released from the liquid surface of plating shop process tanks during plating and anodizing operations. The dirty normal condition scrubber chromic acid removal efficiency is known from the 1987 BAAQMD Test. The best condition properly operated scrubber chromic acid removal efficiency is known from the April 5, April 6, August 10 and August 17 tests. A SECF is needed to correct the amount of chrome measured to have been discharged from freshly cleaned, fully overhauled and properly overhauled scrubbers so that the amount actually discharged from dirty ones during normal operations.
Calculation of this set of two SECFs is easy and straightforward because the measurement of actual scrubber efficiency during all of the source tests in 1987 and 1988 is resistant to any form of cheating and has a very high degree of confidence because of this.
2. Plating Bath Release Correction Factors (BCF): This set of two BCFs is necessary because there was a considerable amount of cheating by Mare Island personnel during the 1988 tests. This cheating was very cleverly accomplished. It's purpose was to reduce the amount of chromic acid actually released from the plating baths themselves when an electrical current was applied to the plating / anodizing baths during the scrubber performance tests. The cheating methods were changed and adjusted from test to test as bad test results came into Mare Island. The basic idea behind the cheating is that the less chromic acid that goes into a scrubber the less will come out. In 1988 the proposed BAAQMD chrome plating regulations were meant to control the amount of chromic acid released from a scrubber and so anything that would decrease the amount of chromic acid released into the scrubber would have a beneficial result insofar as getting new operating permits for the Mare Island plating tanks and scrubbers was concerned..
a. The dismally bad scrubber efficiency results of the 1987 Tests prompted Mare Island to completely rebuild the two chrome scrubbers and to clean and put into the best possible state of repair the remainder of the system. Also, plating bath parameters were adjusted and manipulated to obtain an artificially lowered amount of chromic acid released from the chrome plating and aluminum anodizing baths into the scrubbers during the tests.
b. The results of the April 5 and April 6, 1988 Tests prompted Mare Island to further adjust and manipulate the operating parameters of the chrome plating and aluminum anodizing tanks during the tests. The purpose of this was to further reduce the amount of chromic acid entering the scrubbers so that less would be discharged. Scrubber operating parameters were also slightly manipulated.
H. Calculation of the Scrubber Efficiency Correction Factors (SECF):
The chromic acid removal efficiency of the dirty, worn out, improperly operated chrome scrubbers was determined by the BAAQMD supervised tests performed in 1987. This efficiency is provided in the August 1988 RSA Scrubber Test Report on page 17 to be:
Hard Chrome Scrubber Measured Chromic Acid Removal Efficiency: 17%
Decorative Chrome / Anodizing Scrubber Chromic Acid Removal Efficiency: 7%
The scrubber efficiency measured in the April 5, 1988 BAAQMD Supervised Hard Chrome Plating Test and the April 6, 1988 BAAQMD Supervised Decorative Chrome / Anodizing Scrubber Test is cited in the RSA Source Tests on pages 9 and 11:
Hard Chrome Scrubber Average: 95.7%
Decorative Chrome / Anodizing Scrubber: 93%
Using these efficiencies it is possible to calculate a SECF to correct results from clean scrubbers and convert them to results that would be obtained from normal condition scrubbers. This is a simple calculation and it is the ratio of the dirty scrubber efficiency to the clean scrubber efficiency:
Hard Chrome Scrubber SCF= 95.7% / 17% = 5.63
Decorative Chrome / Anodizing Scrubber SCF = 93% / 7% = 13.29
I. Determination of Hard Chrome Plating Bath Correction Factors (HBCF)
1. Methods of Cheating Used in the Hard Chrome Plating Bath:
a. The test electrical current used during the RSA Test of August 10, 1988 was too low. The RSA Test used test pieces of 18.7 and 16 square feet in the two hard chrome plating baths, both plated simultaneously, using 2000 and 2400 Amperes of electrical current. This totals to 4,400 Amperes for 34.7 square feet of test piece. The Mare Island Plating Shop Process Instruction, Table 1 and Appendices 1 and 7 stipulated that the hard chrome plating electrical current is to be 2.5 Amperes per square inch of material being plated. The test pieces total area is 4996 square inches and so the correct test electrical current can be calculated:
August 10, 1988 Correct Test Electrical Current (Amperes) = 2.5 Amperes per square inch X 4996 square inches = 12,492 Amperes.
Both tanks T-4 and T-5 were connected to independent power supplies with an electrical capacity of 5000 Amperes each for a total of 10,000 Amperes. This calculated value exceeds this and so it also demonstrates that another form of cheating used during the RSA tests was to use test pieces that were too large. It is not possible, with available data, to calculate a correction factor for this at this time.
b. The test current used during the April 5, 1988 test was also too low. In these tests the two same baths were operated at an average total of 8275 Amperes. Assuming the use of the same sized test pieces as were used in the RSA Test, the appropriate normal conditions Amperage should also have been 12,492 Amperes.
Using this data HBCF's can be calculated for the two tests:
April 5, 1988 Test HBCF = 12492 Amperes / 8275 Amperes = 1.51
August 10, 1988 RSA Test HBCF = 12492 / 4400 Amperes = 2.84
Note that once again the results show that after the 1987 test there was a reduction in the Amperage used and then after the April 5, 1988 there was a further reduction for the RSA Test.
This form of cheating was done to artificially reduce the quantity of chromic acid droplets being released from the top of the plating tanks. This was accomplished by using extremely oversized test pieces.
J. Determination of Decorative Chrome Plating and Anodizing Bath Correction Factors (DBCF):
1. Methods of Cheating Used in the Decorative Chrome Plating Bath:
a. Dummying Not Included: The April 6, 1988 test and the RSA August 17 test of the decorative chrome plating bath did not take into account the fact that prior to use the chrome plating bath was to be "dummied" for an hour. This is specified in the Plating Shop Process Instruction , appendix 7, paragraph 1.3 Note. Dummying is necessary with decorative chrome plating. When a plating bath is dummied, a clean piece of metal that is of no value is plated. This is done to remove impurities from the plating bath and to activate the lead anodes that quickly become inactivated when not in use.. During the actual tests the plating bath was operated for four three minute periods per the hour of the test. The test did not measure emissions during the one hour of dummying that had to be accomplished before the bath could be used to plate regular articles for plating. There is no dummying correction factor used for calculations involving hard chrome plating because that test was conducted for a complete hour whereas the decorative chrome plating test was conducted for only 12 minutes out of an hour. If the same size test piece had been used during the dummying conducted for a full hour prior to decorative plating then the emissions from the decorative chrome plating tank would have been much greater. This is obviously why dummying was not included in the test measurements.
The calculation of a dummying correction factor (DCF)is calculated as follows:
Dummying Correction Factor (DCF) =
minutes of dummying / minutes of plating per hour = 60 minutes / 12 minutes = 5
b. Test Current Reduced Below Normal: The amount of electricity (electrical current) passed through a chrome plating bath is measured in units of Amperes. The April 6, test of the decorative chrome plating bath was performed at an electrical current amperage of 1000 Amperes. The same test, performed on the same tank by RSA on August 17, 1988 used 600 Amperes. The question to be answered is either of these the amount of Amperes that would be used during normal operations? The answer is no.
The Mare Island Plating Shop Process Instruction, Table 6 and Appendices 1 and 7 state that the amount of electrical current to be used during chrome plating is 2.5 Amperes per square inch of metal to be plated. The RSA test states that for its test of the decorative chrome plating bath a plating test piece with a size of 7.68 square feet was used. The amount of electrical current that actually would have been used to plate a piece of this size can be calculated:
From this it can easily be seen that during both the April 6, 1988 and the August 17, 1988 RSA tests the electrical current applied to the decorative chrome plating tank was reduced below normal. This data allows the calculation of separate DBCF's for each test:
April 6, 1988 DBCF1 = 2764 amperes / 1000 amperes = 2.76
August 17, 1988 RSA DBCF1 = 2764 amperes / 600 amperes = 4.61
This means simply that Mare Island cheated by using an artificially low electrical current in an attempt to obtain a lower amount of chromic acid discharged from the scrubbers. The 1987 test results made it necessary to make a moderate reduction and when this didn't do the job with the August 6, 1988 tests there was a further reduction for the RSA tests.
Although the April 6, 1988 test document does not identify the size of the metal test piece used there is no reason to suppose a different one was used for both tests. I have assumed in the calculation that the same size test piece (7.68 square feet) was used for both tests.
b. Plating time and Bath Use Was Reduced: The RSA Test of August 17, 1988 states that the decorative chrome plating test consisted of four three minute runs during which a test piece of 7.68 square feet was plated. The electrical current used during each three minute test was 600 Amperes. According to this, in a typical day of two shifts or 14 effective hours,, the decorative plating tank would use electrical current as follows:
Daily Typical Ampere-hours used = 600 Amperes x 14 hours per day x 12 minutes plating per hour / 60 minutes per hour = 1680 Ampere-hours per day.
The question to be answered is this actually normal operating conditions and the answer is no. The Mare Island Toxic Inventory Preview of 1989 shows that for the nine months of 1988 during which the plating shop was operated 780,000 Ampere-hours of electrical current were passed through the decorative chrome plating bath. Adjusting this nine month figure to account for a normal 12 month operating year:
Typical yearly electrical usage = 780,000 Amperes-year X 12 months year / 9 months data = 1,040,000 Ampere-hours
The federal work year is 240 days. The ampere-hours used per day is:
Ampere-hours used per day = 1,040,000 / 240 = 4,333
This demonstrates that the decorative chrome plating conditions during the August 17, 1988 RSA test were not normal but had been considerably reduced to obtain a reduced discharge of chromic acid from the bath. Using this data a DBCF can be calculated for the August 17, 1988 RSA Test:
August 17, 1988 RSA Test DBCF2 = 4,333 Ampere-hours per day actual/ 1680 Ampere-hours per day test = 2.58
There is insufficient data to allow a similar calculation of a DBCF2 for the April 6, 1988 test. However, assuming that all conditions for that test were identical except for the Amperage the following estimation can be calculated:
April 6, 1988 DBCF2 = April 17, 1988 DBCF2 2.58 x 600 Amperes / 1000 Amperes = 1.55
2. Methods of Cheating Used in the Aluminum Anodizing Bath:
Anodizing of aluminum is a different process than chrome plating even though it uses the same chemical, chromic acid, as is used for chrome plating. The two ways that anodizing differs from chrome plating are:
a. In anodizing the piece of aluminum that is anodized is made the anode in the plating bath instead of the cathode whereas during chrome plating this is the opposite. This has a practical effect because the gas that is generated during anodizing on the plate being anodized is oxygen rather than the hydrogen that is generated when chrome plating is being accomplished. The oxygen bubbles generated during anodizing are larger than the hydrogen bubbles and they make larger droplets to be carried into the scrubber. Larger droplets translate to more effective removal in the scrubber.
b. In anodizing it is the Voltage that is to be applied to the aluminum that is being anodized that is specified and not the electrical current in amperes. There is a good reason for this. Chromic acid is an extremely aggressive acid. Anything put into it is immediately begins being decomposed. When a metal piece is being chrome plated, chrome forms on the piece while at the same time chrome is being removed by the action of the chromic acid. Enough electrical current must be supplied during chrome plating so that the deposition rate of chrome is much greater than the removal rate caused by decomposition. In the anodizing process the problem of decomposition is much more severe and because there is no deposition of chromium on the aluminum to protect it, the electricity provided to the piece of aluminum being anodized must be of a sufficient amount to anodize the piece very very quickly. The very thin, hard and tight aluminum oxide film that is formed during anodizing will resist the decomposition effects of the chromic acid only if it is formed very quickly. This is why aluminum anodizing requires a Voltage rather than a current. Current produces deposition in a plating bath and Voltage produces a thin, hard, tightly adherent layer of aluminum oxide in anodizing.
How did Mare Island cheat during the operation of the anodizing tank? The Mare Island Plating Shop Process Instruction, Appendix 2, paragraph 1.3 specifies that anodizing must be conducted for 20 to 50 minutes at 40 Volts. Note that in the April 6, 1988 Test and the RSA August 17, 1988 tests only the Amperes provided to the tank were measured and recorded. The Mare Island Toxic Inventory Preview of 1989 shows that no Amperage was recorded for the anodizing tank and this demonstrates that Amperes were not used as a method of control on this tank. The April 6 test shows the anodizing tank was operated at less than 10 Amperes. The RSA Test on August 17 records that the anodizing tank was operated at 5 Amperes. Once again the amount of electricity was decreased to obtain a better result from the outlet of the scrubber.
The problem is the anodizing Amperage did not matter. The correct parameter to control is Voltage. Recording of Amperage, particularly a very low Amperage is a direct form of cheating. The question that needs to be answered is, what Amperage would result in the anodizing tank if the correct voltage had been applied to it during the tests of April 6 and August 17? The value can be calculated from the base data recorded in the RSA Test in Appendix III. This data shows that when a Voltage of 7.5 Volts was applied to the chromium plating tank with a test piece of 18.7 square feet 2000 Amperes was passed through the tank. It also shows that when a Voltage of 8.5 Volts was applied to the other hard chrome plating tank with a test piece of 16 square feet 2400 Amperes was passed through that tank. The anodizing test was onto a test piece of 1.24 square feet and so using all of this data the Amperage that would be produced in the anodizing tank with an applied Voltage of 40 Volts can be calculated.
There is a simple electrical relationship known as "Ohms Law" This is expressed as
Volts = Amperes / Electrical Resistance (Ohms)
This can be rearranged to obtain a value of Ohms:
Ohms = Amperes / Volts
The Plating Shop Process Instruction Mixing Instructions state that de ionized water is to be used for all plating baths. This means that both chrome plating baths and the anodizing baths will have the same electrical resistance.
For the first tank: Resistance = 2000 Amperes / 7.5 Volts = 266.67 Ohms
For the second tank Resistance = 2400 Amperes / 9.0 Volts = 266.67 Ohms.
But de ionized water is very electrically resistive. It is so resistive that the size of the metal piece in the bath will have a strong effect upon the electrical current produced by a given Voltage. The Ohms value can be calculated per square foot of test piece:
For the first tank: 266.67 Ohms / 18.7 Square feet = 18.14 Ohms per square foot.
For the second tank: 266.67 Ohms / 16 Square feet = 16.67 Ohms per square foot.
These values are close enough to average to 17.40 Ohms per square foot of test piece.
The anodizing tank amperage that would have been produced with a correctly applied voltage of 40 Volts and a test piece of 1.24 square feet size can be calculated:
The Ohm's Law equation is rearranged to produce Amperage from Volts and Resistance:
Volts X Resistance = Amperes
With 40 Volts applied to the anodizing tank with a test piece the sized used during the RSA test, 1.24 square feet, the Amperes passing through the tank would be:
40 Volts X 1.24 square feet x 16.18 Ohms per square feet - 863 Amperes
It can be seen from this that during the April 6 and the August 17 tests the anodizing bath amperage was so small (10 Amperes and 5 Amperes) that there was no emission of chromium acid from the anodizing bath during the April 6 and August 17 tests. It can easily be seen why Mare Island cheated this way. Applying 40 Volts to the anodizing tank would have produced a highly visible chromic acid mist.
Another thing to consider is that the RSA Test report states that the Anodizing test piece was operated at an electrical current of 150 Amperes for one minute and then reduced to 5 Amperes. There is no explanation as to why this was done. This is in complete disagreement with the Mare Island Plating Shop Process Instruction for anodizing of aluminum which states the piece should have been been continually energized at 40 Volts for 20 to 40 minutes. The fact the piece was energized to 150 Amperes for a short time indicates cheating took place.
Note that the concentration of chromic acid in the anodizing bath (75 grams/gallon) is lower than in the plating bath (250 grams/gallon). This means that for the same Amperes passing through an anodizing tank the release of chromic acid will be lower than from the plating bath by the ratio of these different dilutions:
Anodizing Chromic Acid Release = 75 grams/ 250 grams = 0.30 x Decorative Chrome Plating release
These results will be used on the emissions calculation page to estimate normal conditions anodizing bath chromic acid emissions.